Background and overview[1]
2,4,5,6-tetrafluoroisophthalonitrile can be used as a pharmaceutical synthesis intermediate, such as the preparation of dichloruron, which is an insect growth regulator, and the better representative is dichloruron. .
Chemuron has the following advantages: 1. Unique mechanism of action, low toxicity to beneficial organisms, easy to degrade, high selectivity, relatively safe, and is an insect growth regulator. 2. High biological activity and low effective dosage. 3. Has broad-spectrum insecticidal activity. 4. No cross-resistance with other pesticides.
Preparation[1]
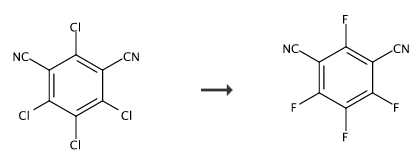
In a 250 ml three-neck flask, add 150 ml of dry sulfolane, and then add 26.6 g of dried chlorothalonil. Under mechanical stirring, raise the temperature to 70°C to completely dissolve it. Quickly add 25.0 g of anhydrous water. Potassium fluoride and 0.5 grams of 18-crown ether-6 are used as phase transfer catalysts, and a drying tube is installed on the side of the tube, and then the temperature is controlled between 160-180°C. After 8 hours of reaction, cool to room temperature and filter. Remove the precipitate (KF+KCl), and wash the above precipitate 2-3 times with 20 ml of sulfolane (the precipitate weighs about 24.9 grams). Combine the filtrate, pour the filtrate into 500 ml of water, stir magnetically for 2 hours, suction filtrate to obtain 17.6 grams of precipitate, and dry Afterwards, cyclohexane was selected for recrystallization to obtain white needle-like crystals weighing approximately 16.4 grams (yield 70.7%). mp: 102-104℃.
Apply[1]
2,4,5,6-Tetrafluoroisophthalonitrile can be used to prepare the volturon intermediate 4,6-diamino-2,5-difluoro-1,3-phthalonitrile: 20 Dissolve grams (0.1 mol) of 2,4,5,6-tetrafluoroisophthalonitrile into 150 grams of 1,2-methoxyethane, and bubble ammonia gas into the solution while stirring within 10 minutes. After the reaction was detected by TLC, it was cooled to room temperature. After cooling, the reaction solution was poured into 150 ml of water and stirred thoroughly. The white solid was obtained by suction filtration, washed with water and dried under vacuum to obtain 17.0 g (yield 87.6%).
References
[1]CN02113831.1 New benzoyl urea compounds and their applications

 微信扫一扫打赏
微信扫一扫打赏

